![The diamagnetic [Ni(CN)_4]^2- ion has square-planar geometry, and the paramagnetic [NiCl_4]^2- ion has tetrahedral geometry. Use crystal field splitting diagrams to explain the difference in the magne | Homework.Study.com The diamagnetic [Ni(CN)_4]^2- ion has square-planar geometry, and the paramagnetic [NiCl_4]^2- ion has tetrahedral geometry. Use crystal field splitting diagrams to explain the difference in the magne | Homework.Study.com](https://homework.study.com/cimages/multimages/16/cms53022462554618608855.jpg)
The diamagnetic [Ni(CN)_4]^2- ion has square-planar geometry, and the paramagnetic [NiCl_4]^2- ion has tetrahedral geometry. Use crystal field splitting diagrams to explain the difference in the magne | Homework.Study.com
Ni(CN)4]^2- is diamagnetic, while [Ni(CN)4]^2- is paramagnetic, explain using crystal field theory. - Sarthaks eConnect | Largest Online Education Community
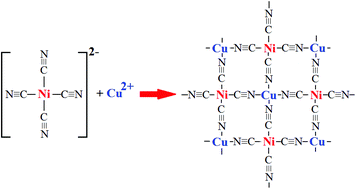
Chemistry and structure by design: ordered CuNi(CN)4 sheets with copper(ii) in a square-planar environment - Dalton Transactions (RSC Publishing)
![Diamagnetic - Paramagnetic - Ni(CO)4, [Ni(CN)4]2- and [NiCl4]2- IIT JEE ... | Chemistry lessons, Chemistry, Lesson Diamagnetic - Paramagnetic - Ni(CO)4, [Ni(CN)4]2- and [NiCl4]2- IIT JEE ... | Chemistry lessons, Chemistry, Lesson](https://i.pinimg.com/originals/a2/20/10/a22010863c43298057a81f0eb09dd692.jpg)
Diamagnetic - Paramagnetic - Ni(CO)4, [Ni(CN)4]2- and [NiCl4]2- IIT JEE ... | Chemistry lessons, Chemistry, Lesson
![Photographs of the M( pz)[M'(CN) 4 ]·2H 2 O powders (M = Ni II , Co II... | Download Scientific Diagram Photographs of the M( pz)[M'(CN) 4 ]·2H 2 O powders (M = Ni II , Co II... | Download Scientific Diagram](https://www.researchgate.net/publication/283244109/figure/fig1/AS:355110174642179@1461676271493/Photographs-of-the-M-pzMCN-4-2H-2-O-powders-M-Ni-II-Co-II-and-M-Ni-II.png)

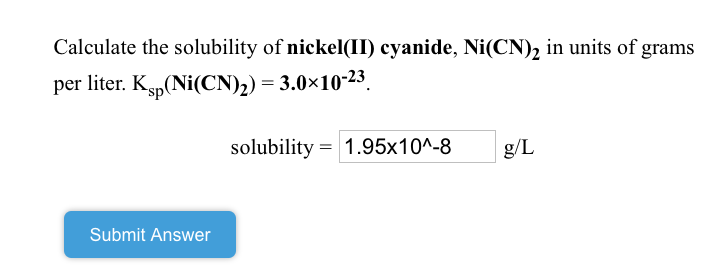
![The geometry of [Ni(CN)4]2− and [NiCl4]2− ions are : The geometry of [Ni(CN)4]2− and [NiCl4]2− ions are :](https://search-static.byjusweb.com/question-images/toppr_ext/questions/345819_212776_ans_5a2a220d536a4adebbfad43e7ddf74b5.png)
![What is the hybridization for [NiCN4]2 ? What is the hybridization for [NiCN4]2 ?](https://byjus-answer-creation.s3.amazonaws.com/uploads/2.14.jpg_img_upload_solution_2022-05-30%2005:07:29.453226.png)
![What is the hybridisation for [Ni(CN) 4] 2-? - Quora What is the hybridisation for [Ni(CN) 4] 2-? - Quora](https://qph.cf2.quoracdn.net/main-qimg-20bb619759136ec1b4419262dd5a28a0.webp)

![Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure-Parmagnetic-Diamagnetic-Examples-dsp2 Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure-Parmagnetic-Diamagnetic-Examples-dsp2](http://www.adichemistry.com/jee/qb/coordination-chemistry/1/q1-2.png)
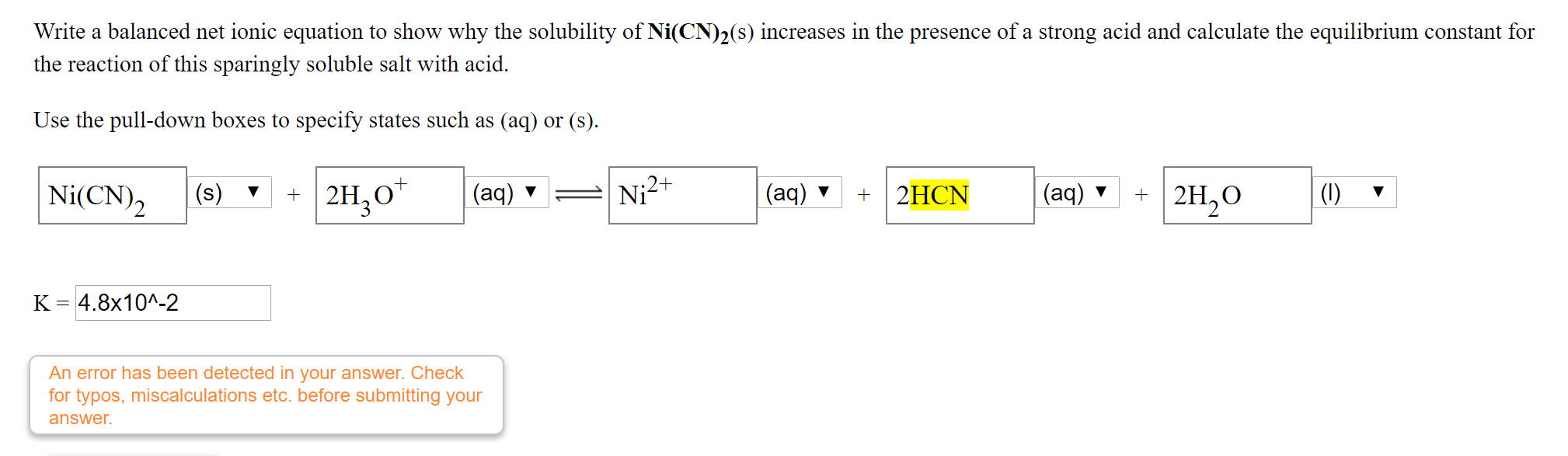

![Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure-Parmagnetic-Diamagnetic-Examples-dsp2 Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure-Parmagnetic-Diamagnetic-Examples-dsp2](http://www.adichemistry.com/jee/qb/coordination-chemistry/1/q1-1.png)
![Ni(CN)4]2- - Cyanonickelate has D4h symmetry Ni(CN)4]2- - Cyanonickelate has D4h symmetry](https://www.chemtube3d.com/images/gallery/inorganicsjpgs/Ni(CN)42-.png)
![Ni(CN)4]2-Hybridisation , Geometry and Magnetic nature -coordination compounds - YouTube Ni(CN)4]2-Hybridisation , Geometry and Magnetic nature -coordination compounds - YouTube](https://i.ytimg.com/vi/5S_my6-2Vkc/maxresdefault.jpg)
