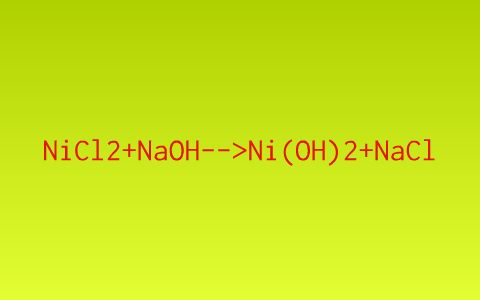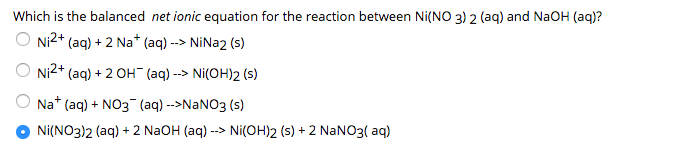5 Six methods of preparing Ni(OH) 2. (a) Basification of a nickel(II)... | Download Scientific Diagram

Calculate the molar solubility of Ni(OH)2 in 0.10 M NaOH solution. The ionic product of Ni(OH)2 2.0 × 10^-15
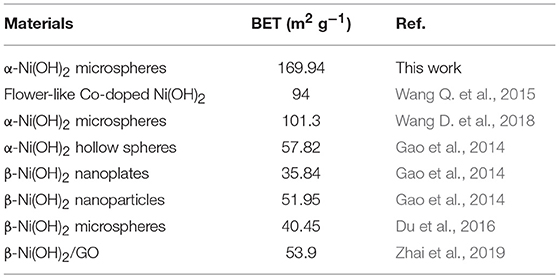
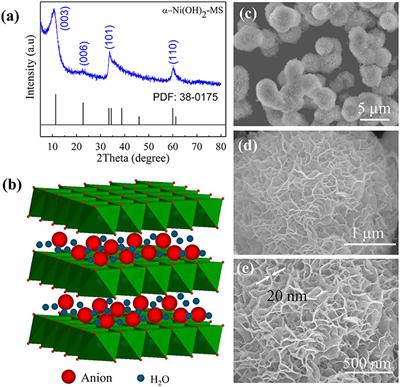
Frontiers | Facile Synthesis of Monodispersed α-Ni(OH)2 Microspheres Assembled by Ultrathin Nanosheets and Its Performance for Oxygen Evolution Reduction

Higher-Valent Nickel Oxides with Improved Oxygen Evolution Activity and Stability in Alkaline Media Prepared by High-Temperature Treatment of Ni(OH) 2 | ACS Catalysis

Calculate the molar solubility of Ni(OH)2 in 0.10M NaOH. The ionic product of Ni(OH)2 is..... - YouTube

One material, multiple functions: graphene/Ni(OH)2 thin films applied in batteries, electrochromism and sensors | Scientific Reports

STA curves: (A) DSC and (B) TGA for reflux-Ni(OH)2 and RT NaOH-Ni(OH)2.... | Download Scientific Diagram

Synthesis of Hexagonal Nickel Hydroxide Nanosheets by Exfoliation of Layered Nickel Hydroxide Intercalated with Dodecyl Sulfate Ions | Journal of the American Chemical Society

NEET 2020 SOLUTION -Find out the solubility of Ni(OH)2 in 0.1 M NaOH . Given that the ionic product - YouTube

QUESTIONFind out the solubility of Ni(OH) 2 in 0.1 M NaOH. Given that the ionic product of Ni(OH)2 is 2×10 - Brainly.in

Figure 5 from Thermodynamic model of Ni(II) solubility, hydrolysis and complex formation with ISA | Semantic Scholar

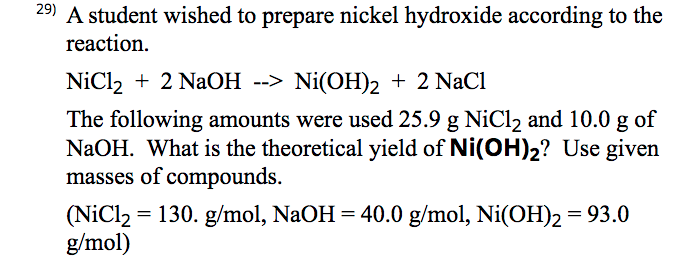

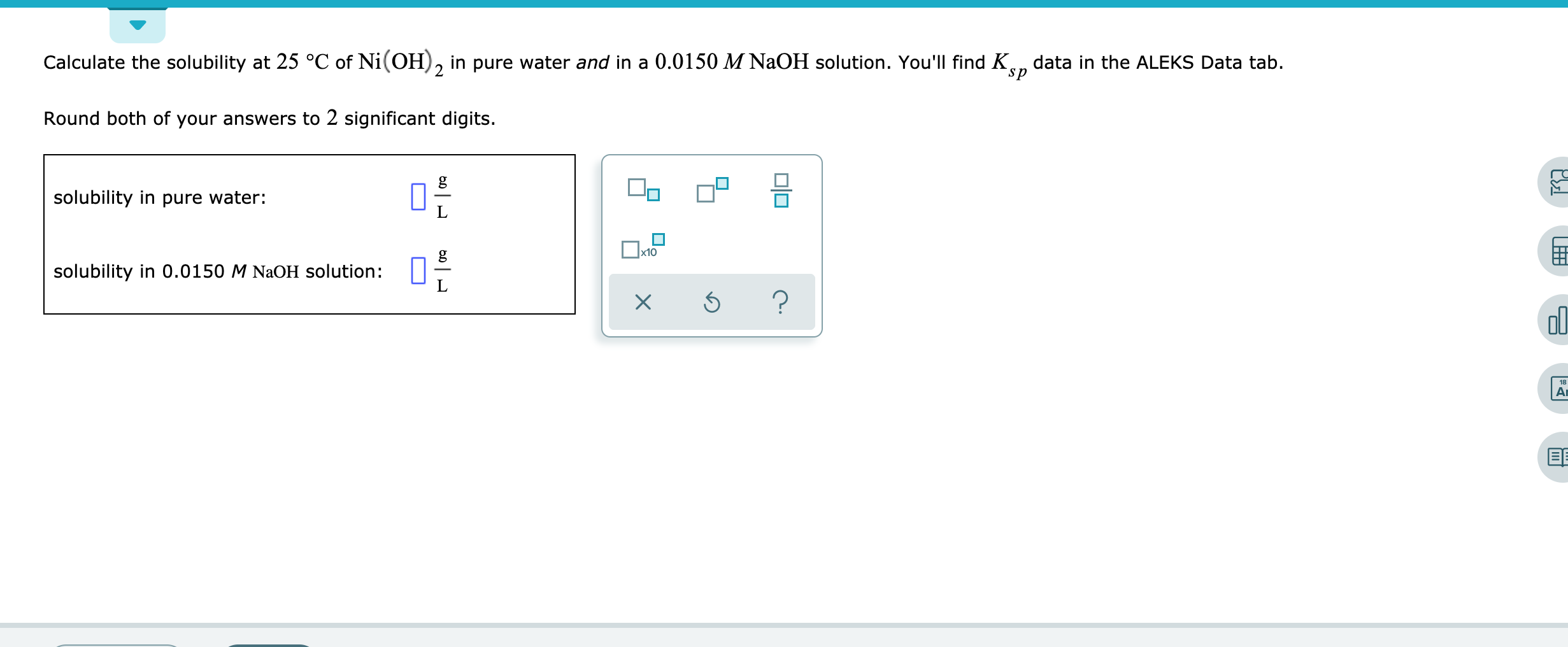



![Malayalam] Calculate the solubility of Ni(OH)2 in 0.1M NaOH solution. Malayalam] Calculate the solubility of Ni(OH)2 in 0.1M NaOH solution.](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/6531163.webp)